The voltage of a single lithium-ion cell (3 to 4V) is too low to supply high power applications. Therefore, it is necessary to assemble cells together to elevate the voltage and be able to deliver sufficient power to the application.
Cells are connected in series (voltage increase) and in parallel (capacity and current increase) to form batteries. Beside, a battery system must integrate the following functions:
- Ensure that the cells are always used in their specified range;
- Ensure user safety
- Inform the user of the battery state (remaining energy, available power, safety)
- Resist and protect the cells against environmental constraints (vibrations, shocks, immersion, weather, salt spray, electromagnetic radiations, fire…).
Therefore, the elements composing a battery system must fulfill the following requirements:
Electronic Battery Management System
The voltage of each cell composing the battery, their temperature and current have to be continuously watched to stay in the range specified by the manufacturer. For example, it is generally forbidden to charge a lithium-ion cell when its temperature is below 0°C. A battery has also to protect itself when its temperature or voltage rise too much (for example above 60°C or 4,2V per cell). The Battery Management System (BMS) continuously overwatches these variables to inform the user about battery state or to protect the battery by disconnecting it. The BMS communicates to the user the remaining energy and the power that the battery can deliver or receive. Some BMS can also estimate the aging of the battery.
Let’s note here that the authorized cell functioning range by the BMS (voltage, temperature and current) have a significant influence on the battery life. It has therefore to be chosen in an appropriate manner to cover the application requirement and ensure the maximum life.
Electric protection
Single cells voltage are not dangerous. But connecting a high number of cells together can lead to dangerous voltage. The battery design has to be adapted to ensure the safety of all user and protect them from the dangers of high voltage. If a battery is designed for a working voltage of 800 V, all current carriers have to be isolated from the chassis for equivalent voltage, with a safety margin generally fixed at twice the nominal voltage added to 1 000 V (e.g. 2 600 V for a 800 V battery). The battery designer has to ensure that all current carriers are separated from the chassis by isolating materials with a sufficient thickness and avoid all possible current leaks around these isolating parts. For example, clean air can ensure a dielectric strength of 3 kV per mm (less with dust). But a current leak path along an isolating part only protects 1,5kV per mm (always in clean air).
Environmental constraints (shock, vibration, EMC, water protection, fire…)
Like every component, the battery will undergo a high number of environmental constraints. Its design and assembly process must be adapted to ensure resistance to these constraints for its whole life. Lithium-ion cells are not designed to resist all constraints of all application. The battery system has therefore to protect the cells from these constraints.
Thermal conditioning and safety
The thermal conditioning of the battery can be necessary to keep the cells in a safe working area. Indeed, between 75°C and 110°C depending on the cell type, cells can go into thermal runaway. Thermal runaway corresponds to a rapid increase of cell temperature followed by a vent opening to evacuate excess of gas or in worst cases to fire or explosion. To avoid these situations, cells must be kept below 55 to 60°C. Lithium-ion cells are designed to keep their integrity during a thermal runaway and evacuate the excess of gas through a vent. The battery system has to be designed to evacuate this high quantity of gas and avoid propagation of thermal runaway to adjacent cells.
Furthermore, a repeated use at high temperature (>35°C) or low temperature (<5°C) cause faster aging of the cells. A study conducted by Renault and The University of Bordeaux (Journal of Power Sources 325 (2016) pages 273 to 285) shows that the aging speed is multiplied by 3,25 between 30 and 45°C. It becomes even more significant between 30 and 60°C, since the aging speed gets multiplied by 21,5.
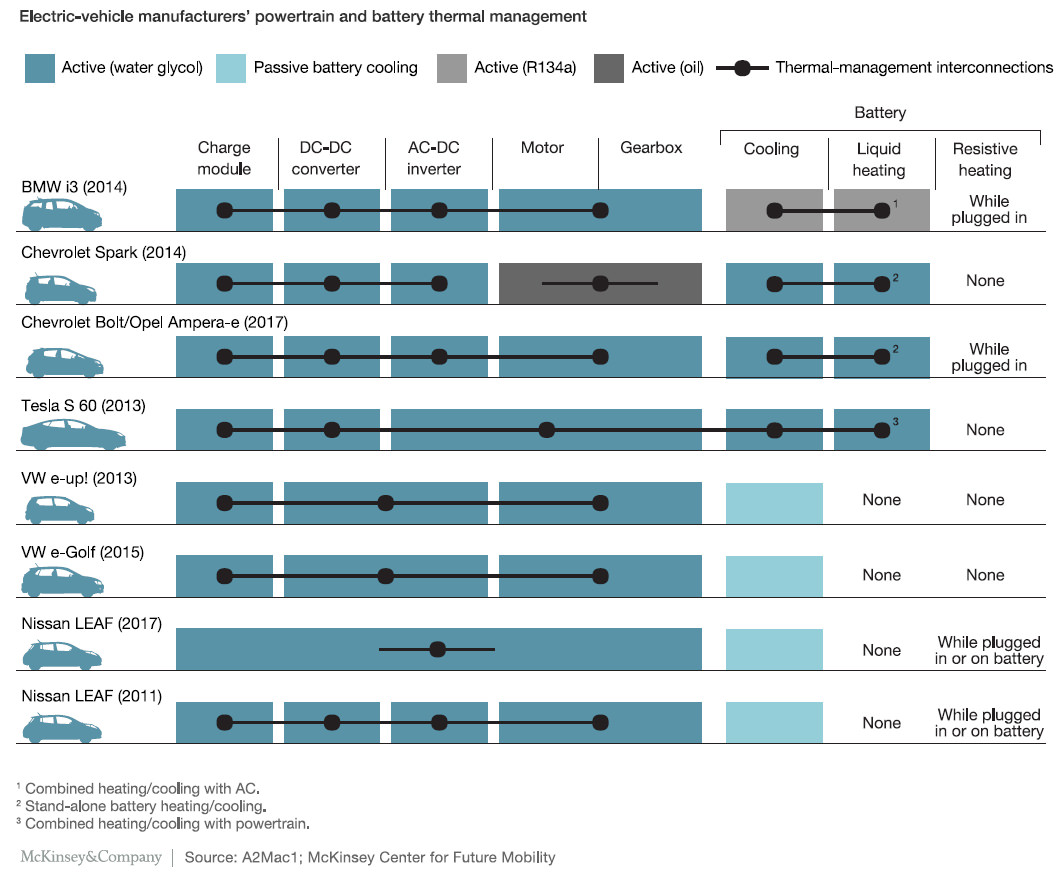
The performance of the battery is always limited by the weakest of its elements, it is then very important to ensure that the cell temperature is as homogeneous as possible to get an identical aging. This lead to the implementation of liquid cooling in numerous electric vehicles (see figure here below).